Novel FcγR humanized mouse models enabling efficacy and PK/PD of therapeutic antibodies
FcγR mouse models for efficacy and PK/PD
Abstract
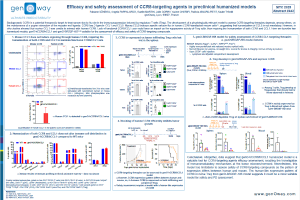
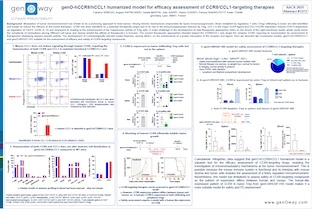
Therapeutic antibodies have revolutionized the way we treat cancer. Enhanced activity of therapeutic IgG can be achieved by the modulation of Fc binding to Fcγ receptors (FcγR), which will consequently modulate the Fc-effector functions triggered upon crosslinking of target and effector cells by these antibodies. Assessment of therapeutic antibodies pharmacodynamic (PD) in preclinical models is challenging, as the FcγR are different from mice to human, as well as their expression pattern. FcRn is involved in the recycling and transport of IgG and is a key determinant of the pharmacokinetics (PK) of IgG. We report here a novel mouse model expressing humanized FcγR with a human-like expression pattern, in which humanized FcγRI, FcγRIIA, FcγRIIB, FcγRIIIA, FcγRIIIB are expressed and replace their mouse orthologs. The model was intercrossed with the hSA/hFcRn mouse model (Viuff et al., 2016), which has shown to improve translatability of PK assessment of therapeutics with extended half-life via FcRn recycling and/or HSA binding. Investigation of FcγRI, FcγRIIA, FcγRIIB, FcγRIIIA, FcγRIIIB expression on DC, monocytes, granulocytes, NK, T and B cells shows that expression pattern in hFcγR mice is consistent with their expression on human PBMC, with a few exceptions. These receptors are functional, as a human IgG1 targeting mouse CD20 induces approximately 50% of B-cell depletion in blood and liver, after a single iv administration in humanized FcγR (hFcγR) mice. This depletion is Fc-mediated, as it is abrogated when hFcγR mice are treated with an anti-mouse CD20 human IgG1 with a Fc-null mutation. Assessment of antibody-dependent cell cytotoxicity (ADCC) ex vivo to investigate FcγR functionality in NK cells shows that Rituximab, an anti-human CD20 chimeric IgG1 with regular Fc, induces higher ADCC efficacy in NK cells from hFcγR than in NK cells from WT mice. Obinutuzumab, an anti-human CD20 featuring an optimized Fc portion to enhance binding to FcγRIII, shows higher tumor cell lysis than Rituximab in NK cells from hFcγR mice, suggesting this model enables ranking of antibodies. Moreover, assessment of activity of Fc-engineered anti-influenza IgG antibodies also shows that anti-Flu antibody with enhanced Fc binding outperforms the WT antibody, improves protective activity, and demonstrates effector function in influenza challenge model, following prophylactic treatment. Conversely, hFcγR mice treated with an Fc-null version of the anti-Flu antibody shows reduced protective activity and survival compared with the WT antibody. Therefore, the model reported here is a novel tool for testing efficacy of Fc-engineered therapeutic antibodies. We are currently investigating the PK profile of therapeutic antibodies in the hFcγR/hSA/hFcRn model. This novel tool is being improved to enable tolerability to human IgG1, biodistribution to the central nervous system, and flexibility of test of therapeutics through expression of human immune checkpoints.

Scientific excellence
From model design to experimental results
Featured in 600+ scientific articles

Collaborative approach
Collaboration with 17 Top Pharmas,
170+ Biotechs and 380+ Academic Institutions

Robust validation data on catalog models
Generated with biopharma partners and in-house

Innovative technologies
and guaranteed freedom to operate

Easy access to models
Models with certified health status from professional breeders in US and Europe