Applications of the genO‑hSA/hFcRn mouse model
Broad range of applications in the fields of immuno-oncology, infectious diseases and autoimmunity:
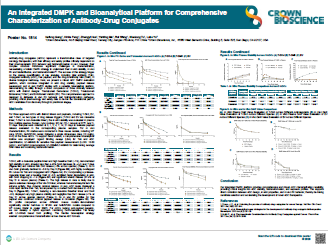
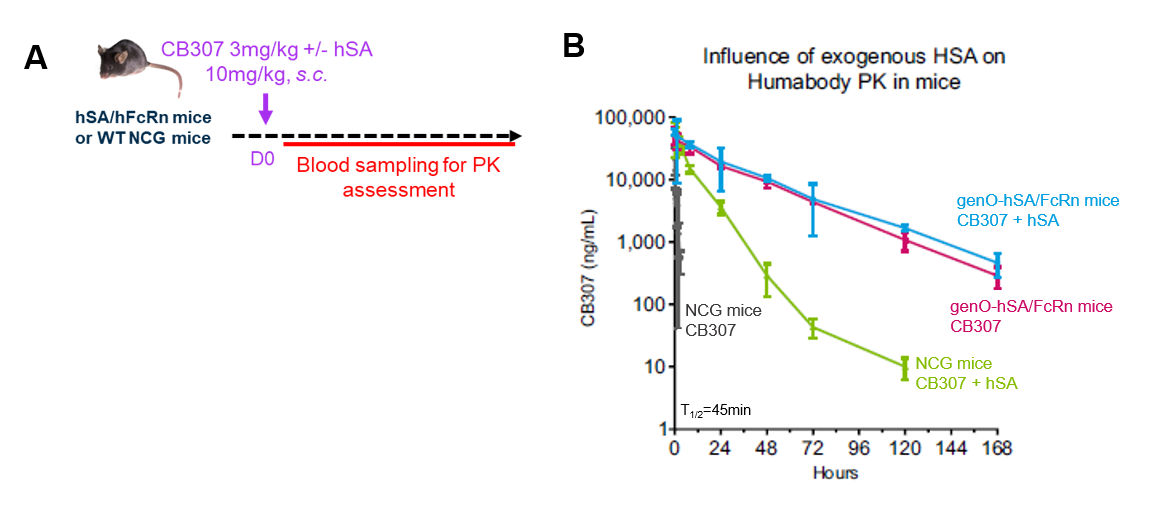
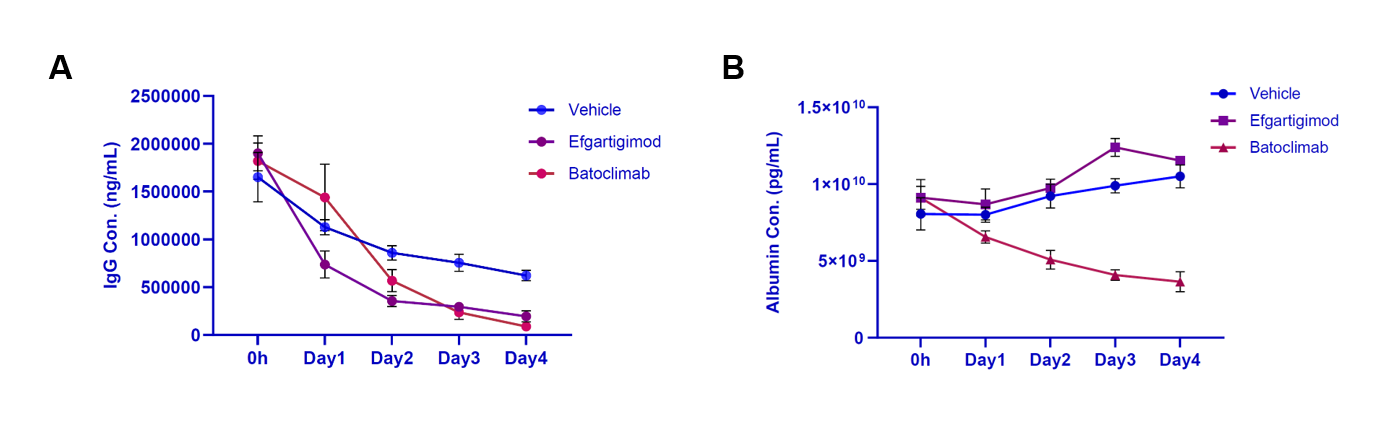
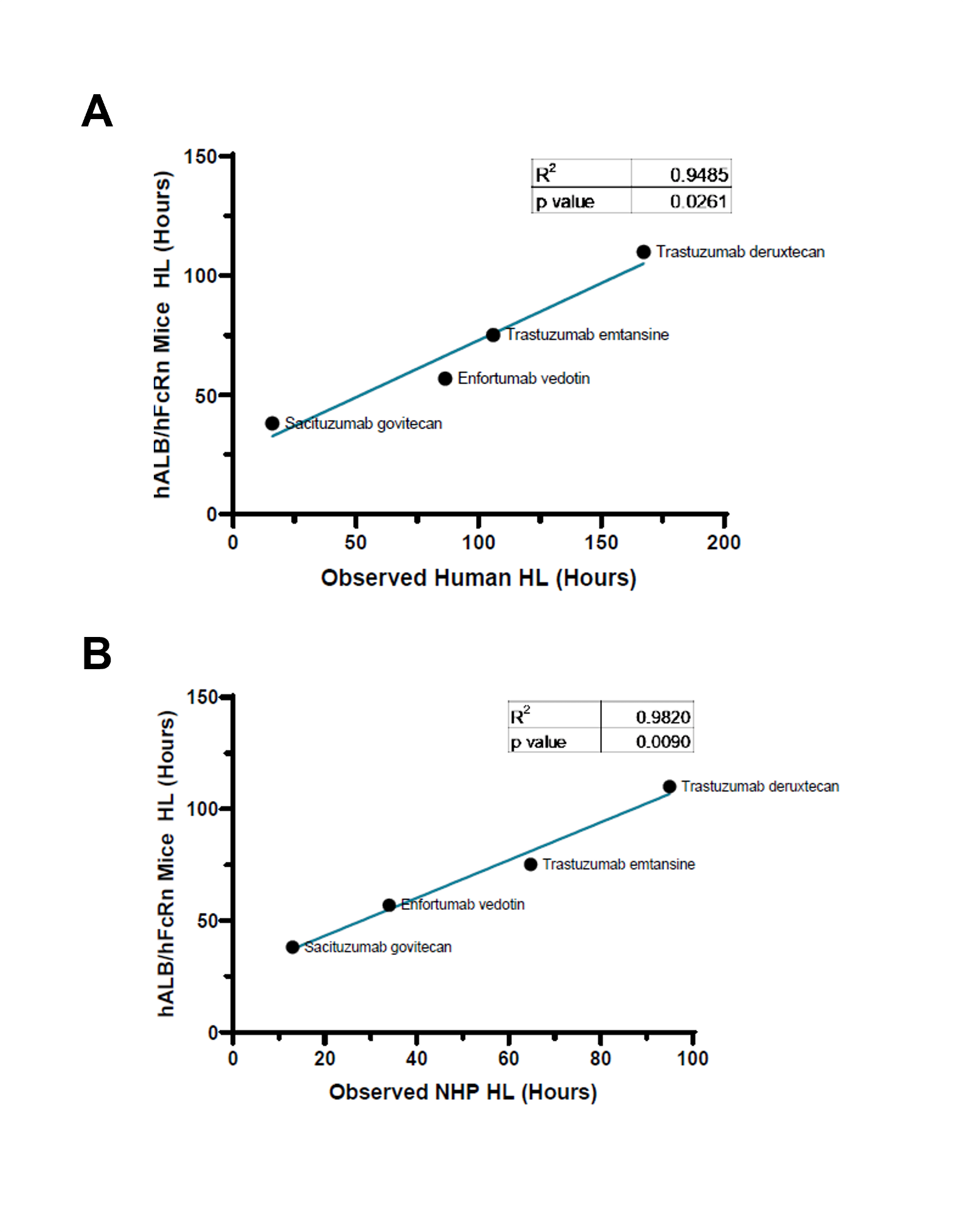
- Assess pharmacokinetics and pharmacodynamics (PK/PD) features, including biodistribution and binding affinity
- Screen compounds based on extended half-life
- Assess the efficacy of optimized therapeutics such as Fc-engineered antibodies, constructs fused to hSA, hSA- and/or FcRn-binding proteins or small molecules
Features
- Physiological expression and distribution of hSA and human FcRn
- Lack of expression of the murine target genes
- Higher physiological affinity of human serum albumin to human FcRn
- Unaltered blood chemistry compared to a wild-type mouse
Guide: Compare FcRn mouse models and identify the best fit for your antibody PK/PD studies. Read the guide →
Clients




.png)