Bridging the species gap in FcγR biology for a translational assessment of therapeutic antibodies in mouse models

Van Damme et al., Cross-species cellular mapping and humanization of Fcγ receptors to advance antibody modeling, Science Immunology, 2026
Therapeutic antibodies rely on their Fc domain to engage Fcγ receptors (FcγRs) and FcRn, shaping effector functions, tissue distribution and half‑life1-3. However, mouse and macaque FcγR repertoires differ substantially from those of humans in gene content, affinity hierarchy, cell‑type expression and polymorphisms2,4,5. As a result, classical murine and non‑human primate (NHP) models often fail to predict human FcγR‑mediated efficacy or safety.
Fc as a primary driver for ranking and optimizing antibodies
For many IgG based therapeutics, efficacy is shaped by the balance between activating and inhibitory FcγRs, which dictates the strength and direction of the immune response, whereas FcRn controls antibody persistence by regulating IgG recycling and serum half life1,3,6,7. However, even subtle Fc alterations, such as changes in glycosylation or engineered mutations, can profoundly disrupt these receptor interactions, creating major challenges for therapeutic development1,3,5,6. These small modifications can unpredictably increase or diminish effector functions, reduce FcγR engagement required for mechanisms such as ADCC or ADCP, or conversely enhance binding to inhibitory receptors like FcγRIIb, altering therapeutic potency. They can also impact clearance rates through altered FcRn affinity. Understanding these mechanisms is essential for dose selection and safety assessments. Consequently, to reliably assess their antibodies, researchers must rely on mouse models that recapitulate human FcγR and FcRn biology.
Limitations of classical mouse and NHP models
Recent cross species analyses show that humans, macaques and mice differ profoundly in FcγR and FcRn expression across immune and non-immune tissues, with mismatches in receptor diversity, cytokine responsiveness and transcriptional regulation10. Mice also lack key human receptors (e.g., FcγRIIA, FcγRIIIB) and display distinct FcγRIII/FcγRIIB distribution on myeloid and B cells2,4. NHPs resemble humans more closely in their IgG subclasses, but they still show important differences in FcγR expression, such as the absence of FcγRIIIB, and polymorphisms, and their use is constrained by cost and ethical considerations5,8. Species specific FcRn biology further affects IgG recycling and PK/PD translation7-9. Collectively, these discrepancies explain why mouse and NHP models frequently mispredict human Fc dependent immune functions.
Current strategies to overcome the translational gap
To address these issues, researchers initially rely on human cell-based FcγR assays such as FcγR reporter lines and primary NK cells, monocytes and macrophages to quantify FcγR dependent functions (ADCC, ADCP, cytokine release)3,6. These in vitro assays are essential for dissecting receptor specific activity but they do not capture whole organism IgG distribution or FcRn regulated IgG recycling. In parallel, human FcRn transgenic mice have been developed to improve pharmacokinetic (PK) prediction. Although these models more accurately simulate human IgG recycling and serum persistence8, they still contain murine FcγRs, which limits their use for predicting the Fc-dependent efficacy or safety of antibodies. This has driven the development of mice humanized for the complete FcγR repertoire, ideally combined with human FcRn.
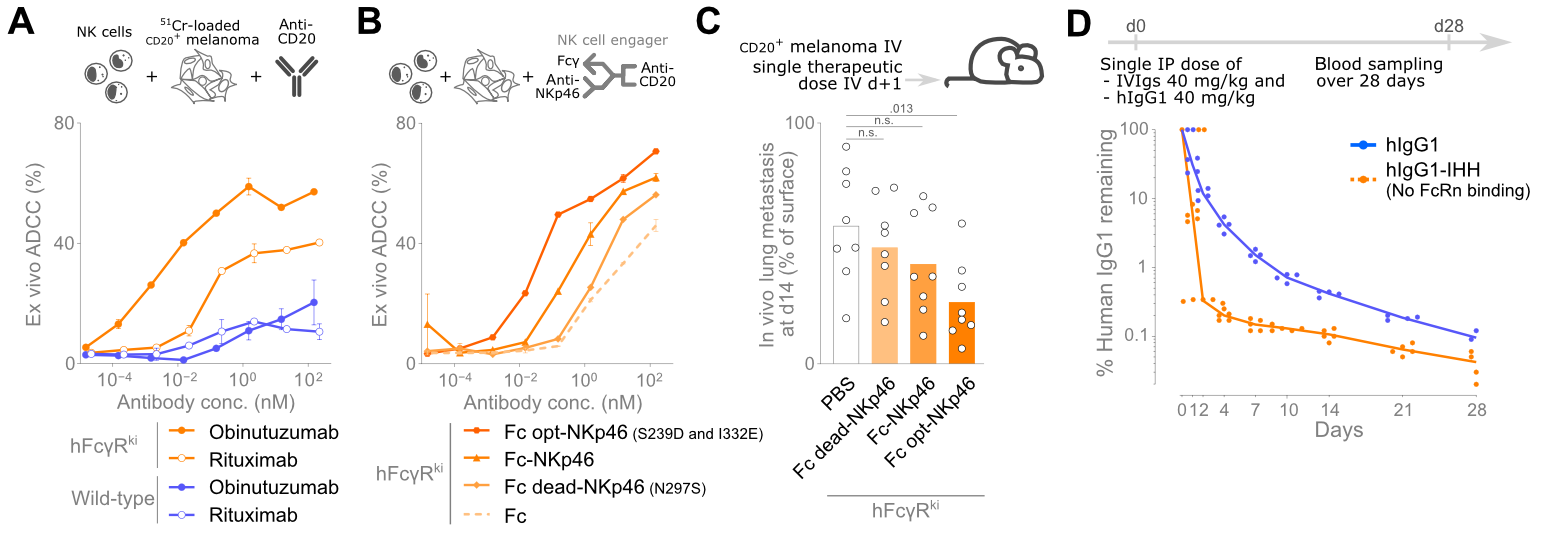
To solve the problem of species-differences in FcγR expression, genOway developed two new mouse models, genO-hFcγR and genO-hFcγR/hFcRn, which express fully functional humanized receptors (FcγRI, FcγRIIA, FcγRIIB, FcγRIIIA, FcγRIIIB and hFcRn for the latter) with a human-like expression pattern. To be able to closely mimic human biology, the receptors were kept expressed under control of their human promotors, and their murine counterparts were knocked-out. These models were deeply characterized in a recent study by Van Damme and colleagues (Science Immunology), who showed that these mice reproduce human like receptor diversity, cell type expression, and cytokine driven modulation, enabling accurate in vivo discrimination of Fc engineered antibodies10. Thanks to these features, these models distinguish Fc dead, wild type and Fc enhanced variants (Figure 1) across infection, B cell depletion, NK cell engager activity and FcγRIIA dependent anaphylaxis, as well as modelling PK profiles10. Such findings demonstrate that only models reproducing human FcγR and FcRn biology enable the translational ranking and optimization of therapeutic antibodies before entering the clinic.
References
- Nimmerjahn F, Ravetch JV. Fcγ receptors as regulators of immune responses. Nat Rev Immunol. 2008;8(1):34–47. https://doi.org/10.1038/nri2206
- Bruhns P. Properties of mouse and human IgG receptors and their contribution to disease models. Blood. 2012;119(24):5640–5649. https://doi.org/10.1182/blood-2012-01-380121
- Bournazos, S., Gupta, A. & Ravetch, J.V. The role of IgG Fc receptors in antibody-dependent enhancement. Nat Rev Immunol 20, 633–643 (2020). https://doi.org/10.1038/s41577-020-00410-0
- Bruhns P, Jönsson F. Mouse and human FcR effector functions. Immunol Rev. 2015;268(1):25–51. https://doi.org/10.1111/imr.12350
- Lux A, Yu X, Scanlan CN, Nimmerjahn F, Impact of Immune Complex Size and Glycosylation on IgG Binding to Human FcγRs, The Journal of Immunology 2013, 190 (8), 4315–4323, https://doi.org/10.4049/jimmunol.1200501
- Wang X, Mathieu M, Brezski RJ. IgG Fc engineering to modulate antibody effector functions. Protein Cell. 2018;9(1):63–73. https://doi.org/10.1007/s13238-017-0473-8
- Roopenian DC, Akilesh S. FcRn: the neonatal Fc receptor comes of age. Nat Rev Immunol. 2007;7(9):715–725. https://doi.org/10.1038/nri2155
- Smith P, DiLillo DJ, Bournazos S, Li F, Ravetch JV, Mouse model recapitulating human Fcγ receptor structural and functional diversity, Proc. Natl. Acad. Sci. U.S.A. 2012; 109 (16) 6181-6186, https://doi.org/10.1073/pnas.1203954109
- Beutier H, Gillis CM, Iannascoli B, et al. IgG subclasses determine pathways of anaphylaxis in mice. J Allergy Clin Immunol. 2017 Jan;139(1):269-280.e7. doi: 10.1016/j.jaci.2016.03.028.
- Van Damme KFA, et al. Cross-species cellular mapping and humanization of Fcγ receptors to advance antibody modeling. Sci. Immunol.11,eady7328 (2026). DOI:10.1126/sciimmunol.ady7328
Related products
Catalogue product
Customized product

Scientific excellence
From model design to experimental results
Featured in 600+ scientific articles

Collaborative approach
Collaboration with 17 Top Pharmas,
170+ Biotechs and 380+ Academic Institutions

Robust validation data on catalog models
Generated with biopharma partners and in-house

Innovative technologies
and guaranteed freedom to operate

Easy access to models
Models with certified health status from professional breeders in US and Europe
