Safety assessment of TCE
Predict Cytokine Release Syndrome Risk and Define the Therapeutic Window Before Clinical Studies
Predict Cytokine Release Syndrome Risk and Define the Therapeutic Window Before Clinical Studies
Effective T-cell engagers (TCEs) can transform patient outcomes, but only if you can confidently balance efficacy with safety. Our mouse models enable you to predict cytokine release syndrome (CRS) before clinical studies.
Whether you're developing bispecifics, trispecifics, Fc engineered molecules, or costimulatory CD28 based TCEs, genOway provide human relevant mouse models validated with biopharma partners and TAA-expressing cell lines that help you generate more predictive efficacy data:
Despite strong anti-tumor potential, TCEs can generate severe, dose limiting-toxicity:
To prioritize safe candidates, researchers need preclinical systems that reflect human immune drivers of CRS, not oversimplified models that underpredict risk.
Our mouse models are engineered to help you confidently assess TCE safety, define therapeutic windows, and identify CRS-prone constructs early.
The genO-panhCD3 model provides a robust foundation for TCE safety assessment because it reproduces human CD3 biology with high fidelity:
Guide: Compare CD3 mouse models for T-cell engager efficacy and safety studies. Read the guide →
These features allow for evaluation of TCE-induced activation and cytokine release under conditions that are truly predictive of clinical safety risks, including CRS, as shown in figure 1:
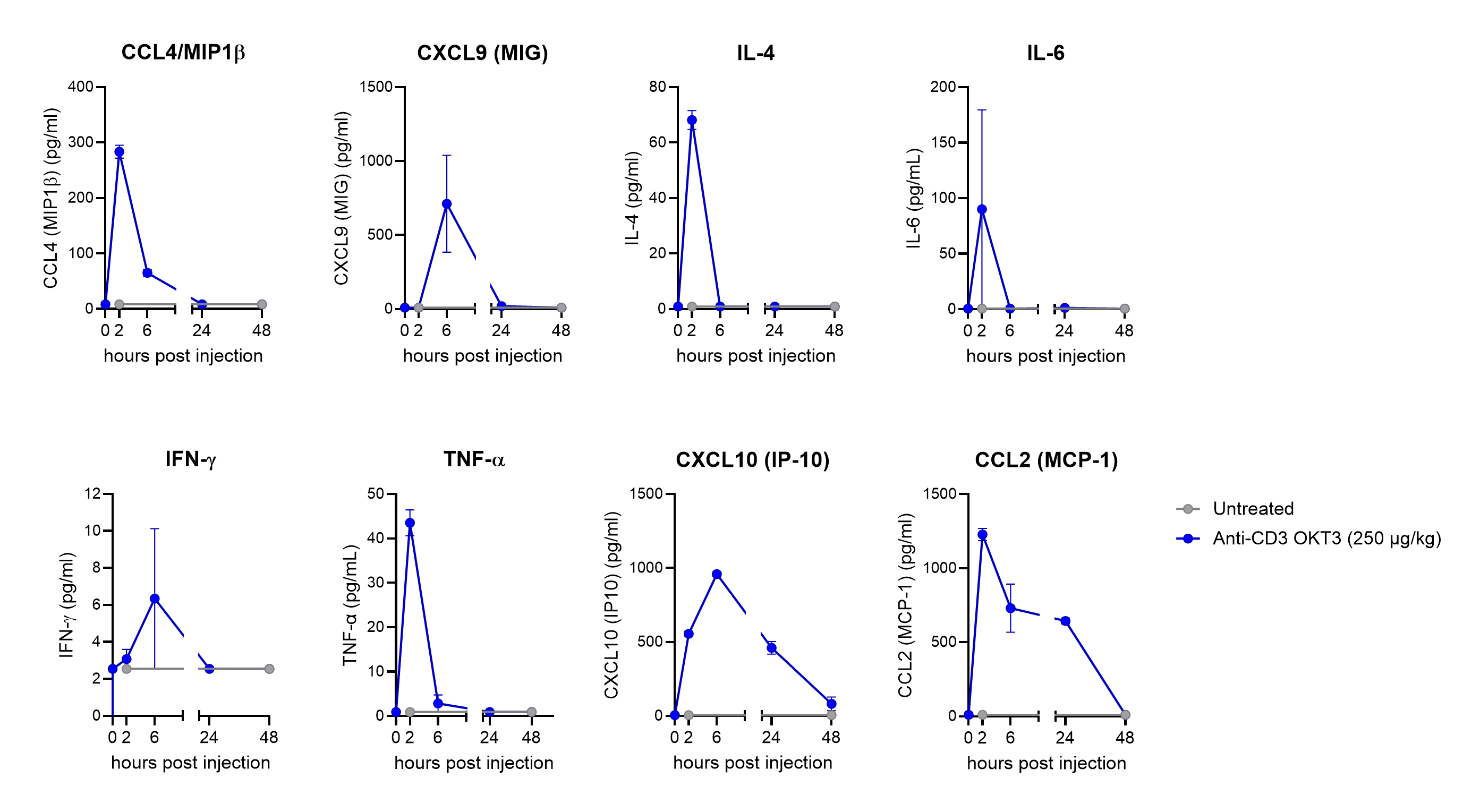
Additionally, this mouse model was also recently used for testing the safety of a next generation TCE targeting CD3 and PD-L1 (SCRI-6), showing improved safety relative to a TCE with the SP34 variant (figure 2):


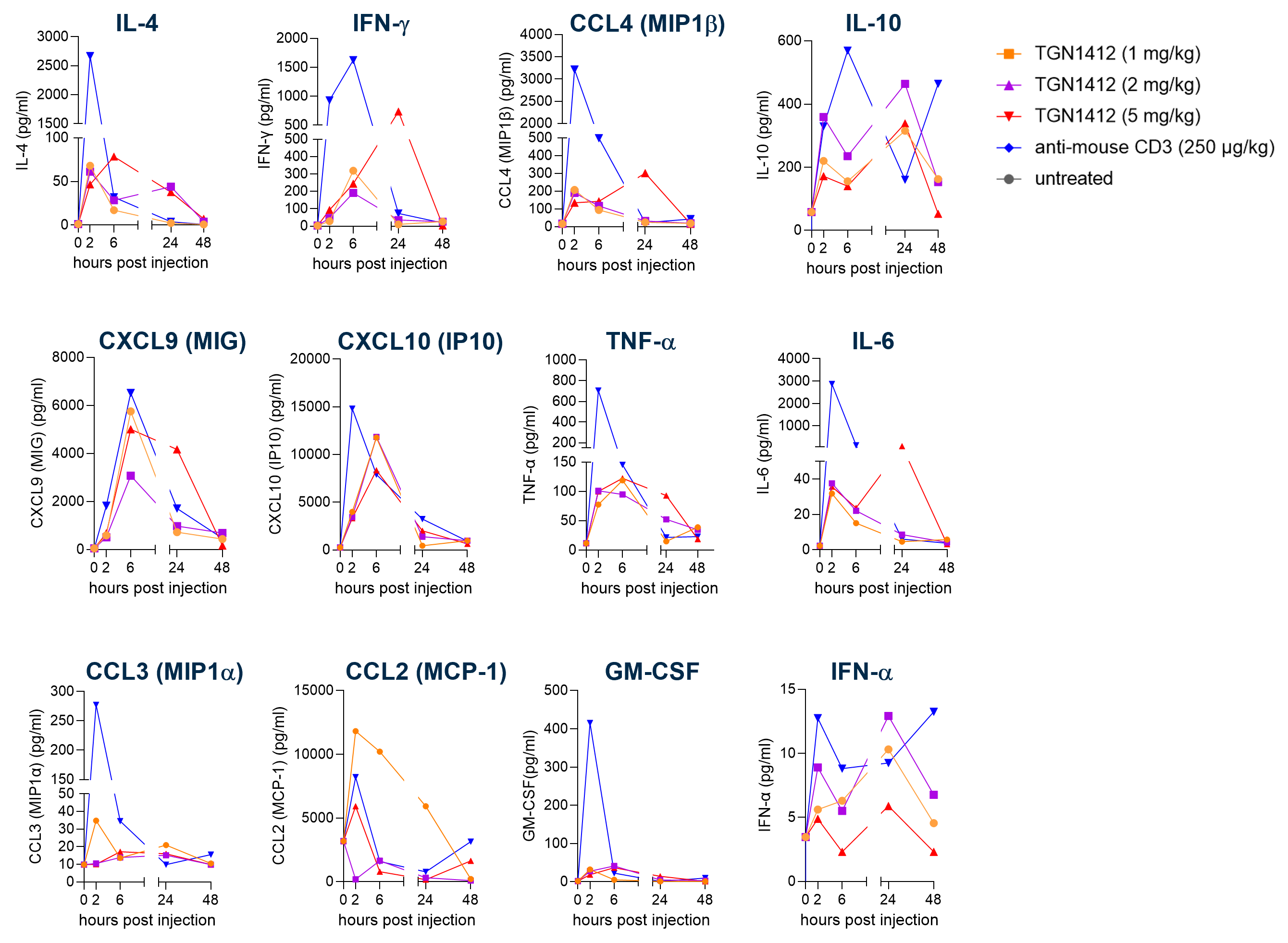
As part of our portfolio of mouse models for evaluating the toxicity of TCEs, we also make available the genO-hCD28 model, which enables the evaluation of next generation TCEs that rely on CD28 co-stimulation thanks to the physiological regulation and expression pattern of hCD28 and the expression of different hCD28 isoforms. This model has been shown to reproduce the CRS-like features induced by TGN1412 as shown in figure 3, as presented at the AACR 2024:

Additionally, we have also developed a mouse model with a human immune system, the genO-BRGSF-HIS, that contains a functional human myeloid and lymphoid cell compartment following reconstitution with CD34+ hematopoietic stem cells. A functional myeloid compartment is essential for measuring CRS-like features since CRS is not driven by T cells alone. Clinical evidence shows that myeloid and dendritic cells play central roles in amplifying cytokine cascades.
This is why models such as the genO-BRGSF-HIS, provide a more reliable foundation for safety evaluation. It demonstrates:
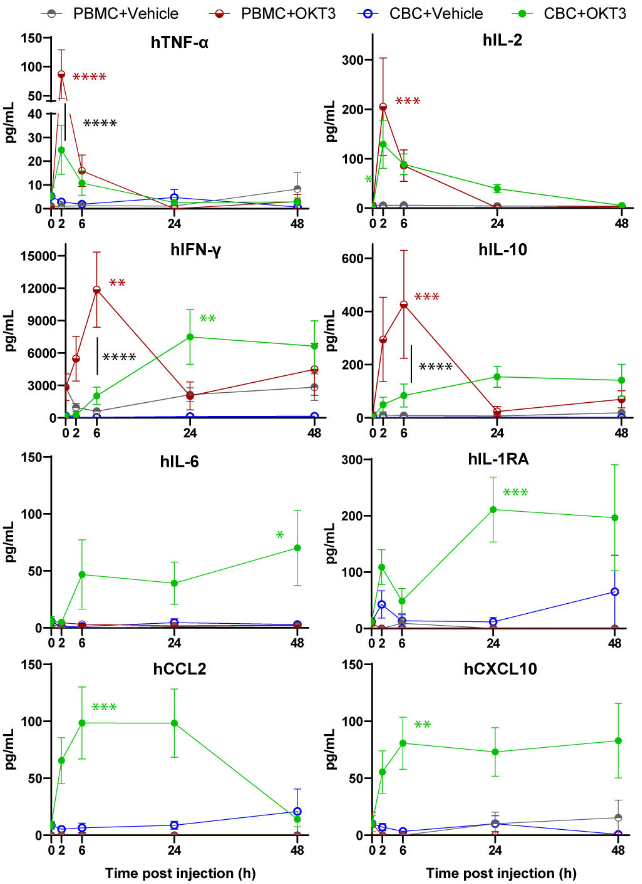
These features make the model particularly suited for understanding how TCE designs translate into CRS propensity, helping teams prioritize safer constructs before submitting their investigational new drug application (IND).
If you’d like to know more about how this model helps you assess the safety of therapeutics, watch our webinar.

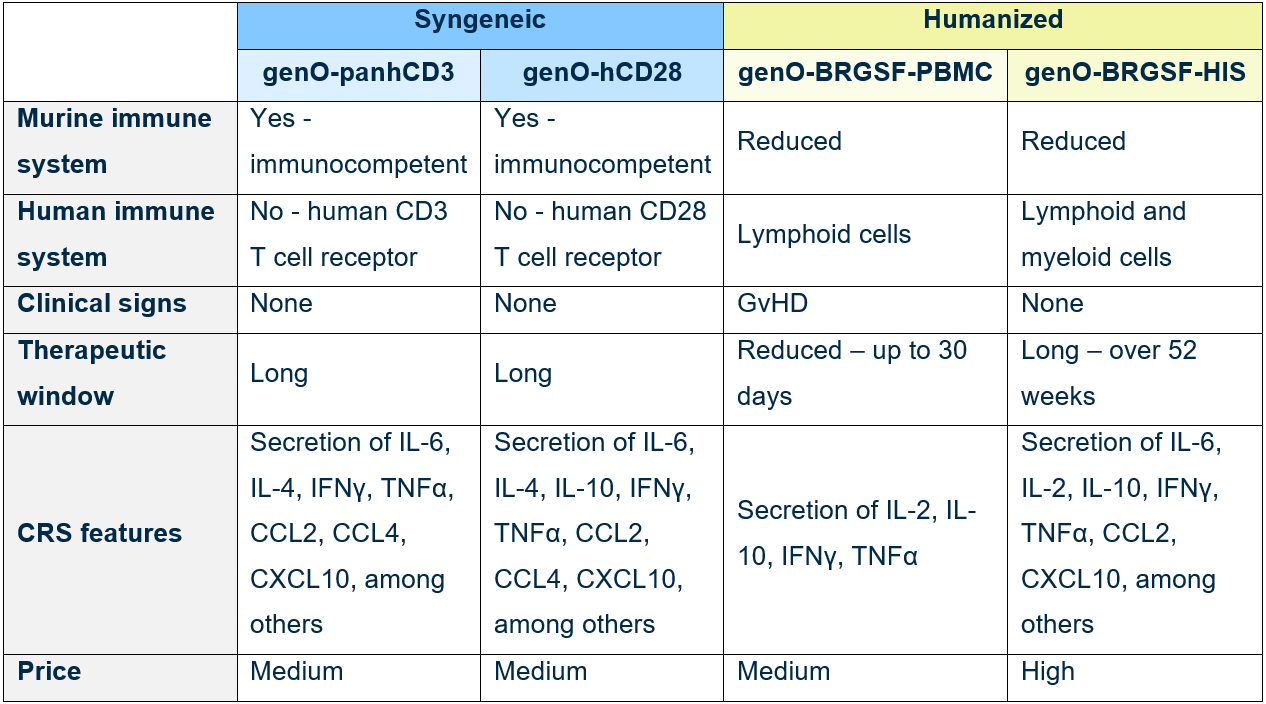
Below you can find a summary of key differences between our mouse models (table 1), which may help you choose the most appropriate model for your study.
Additionally, if you’d like to know more about our model portfolio for TCE assessment, you can find more information here.



From model design to experimental results
Featured in 600+ scientific articles

Collaboration with 17 Top Pharmas,
170+ Biotechs and 380+ Academic Institutions

Generated with biopharma partners and in-house

and guaranteed freedom to operate

Models with certified health status from professional breeders in US and Europe