Efficacy assessment of TCE
More reliable efficacy readouts with pre-clinical models built for T-cell engager biology
More reliable efficacy readouts with pre-clinical models built for T-cell engager biology
Testing T cell engagers (TCEs) for immuno-oncology can be difficult when preclinical models fail to reflect human target expression, T cell activation, and tumor immune evasion mechanisms such as inhibitory checkpoints and immunosuppressive cells. Oversimplified mouse models, surrogate constructs, and PBMC reconstituted systems often overestimate efficacy and fail to predict clinical behavior.
Whether you're developing bispecifics, trispecifics, Fc engineered molecules, or costimulatory CD28-based TCEs, genOway provides human relevant mouse models validated with biopharma partners and TAA-expressing cell lines that help you generate more predictive efficacy data:

genOway’s portfolio of hCD3 models has been used by biopharma companies like AstraZeneca and Pfizer to reliably assess their T-cell engagers. This is thanks to the following features:
These features ensure your TCE engages its target like it would in humans, delivering more reliable efficacy predictions.
Guide: Compare CD3 mouse models for T-cell engager efficacy and safety studies. Read the guide →
Examples of pharmaceutical companies that have used genOway’s hCD3 models
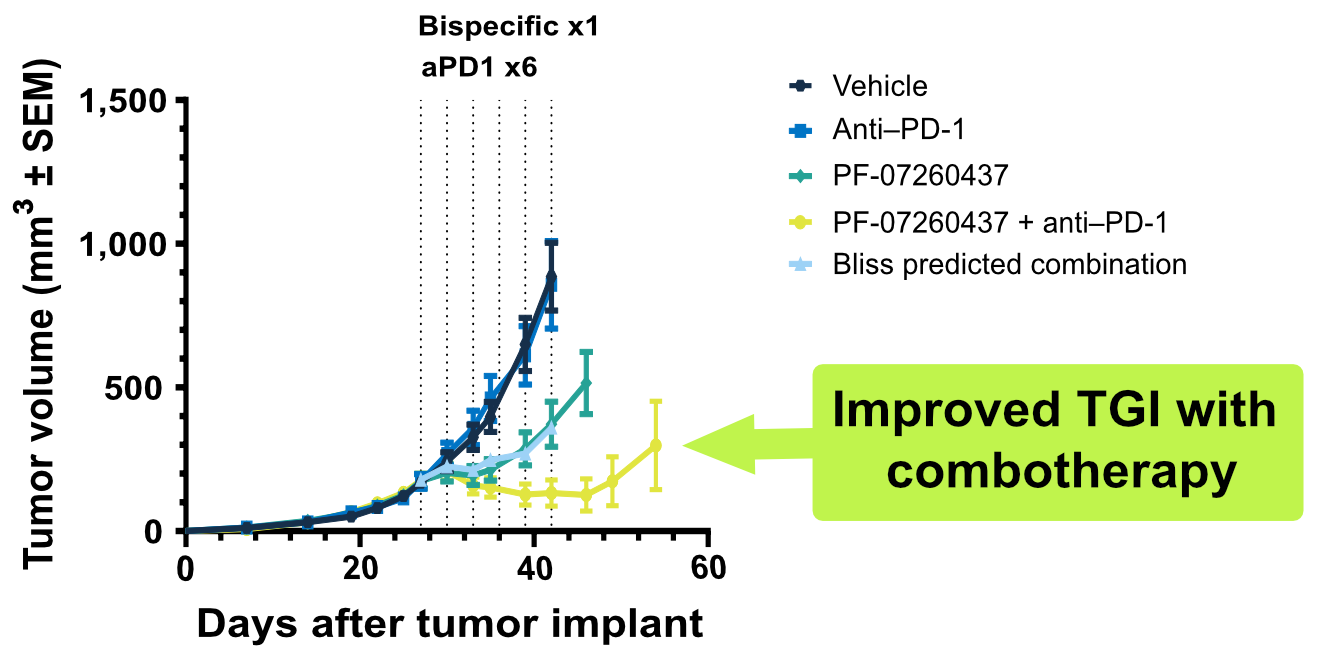
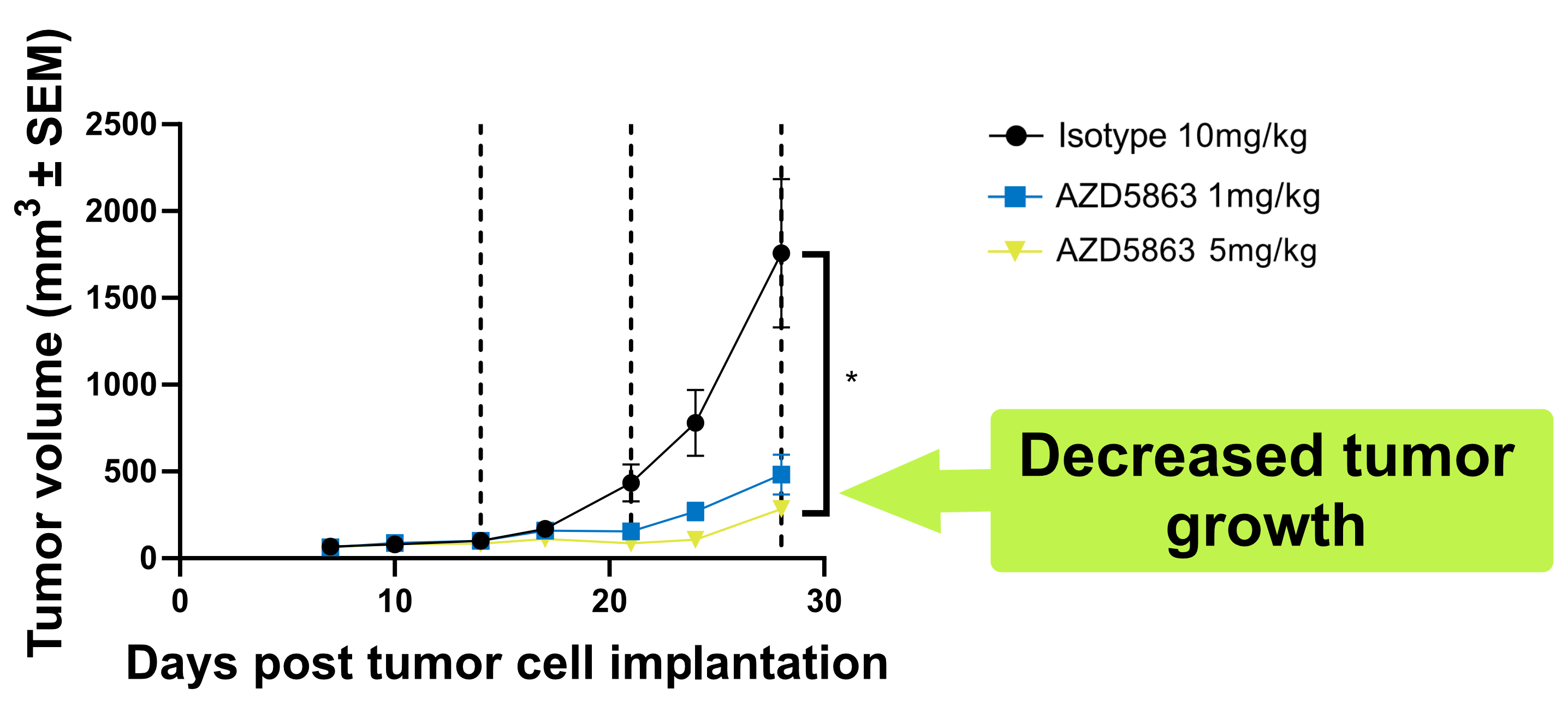
Our hCD3 mouse model portfolio includes options for testing trispecific TCEs, TCEs used in combination with immune checkpoint inhibitors, and TCEs featuring various Fc-engineered regions:
Additionally, if you’d like to know more about our model portfolio for TCE assessment, you can find more information here.

genOway’s hCD28 models (genO-hCD28 and genO-panhCD3/hCD28) allow the evaluation of next‑generation TCEs that rely on CD28 co-stimulation. They display:
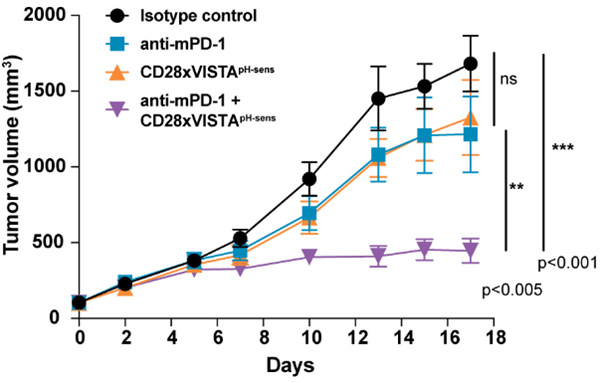
The genO-hCD28 model was validated by Sensei Biotherapeutics, among others, who characterized a tumor-selective bispecific antibody that conditionally co-stimulates T cells via CD28 in a pH- and VISTA-dependent manner:

genOway’s catalog of syngeneic cell lines expressing human TAAs or different human immune checkpoints increases the translatability of efficacy studies by ensuring:
Furthermore, these cell lines can be customized to your specific needs.

The genO-BRGSF-HIS model is a CD34+ HSC reconstituted mouse with a functional human immune system. It captures key tumor microenvironment features thanks to:
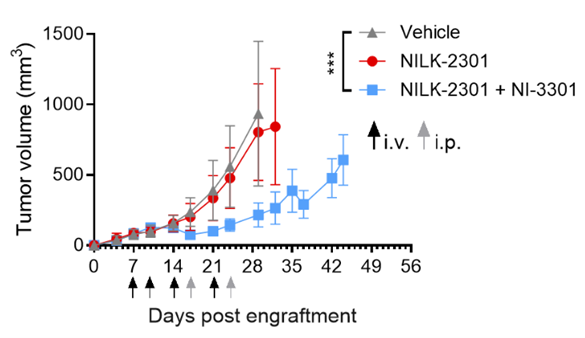
This model recapitulates the tumor-immune dynamics as described in Martin et al., 2025, enabling the translational assessment of TCE efficacy:



From model design to experimental results
Featured in 600+ scientific articles

Collaboration with 17 Top Pharmas,
170+ Biotechs and 380+ Academic Institutions

Generated with biopharma partners and in-house

and guaranteed freedom to operate

Models with certified health status from professional breeders in US and Europe