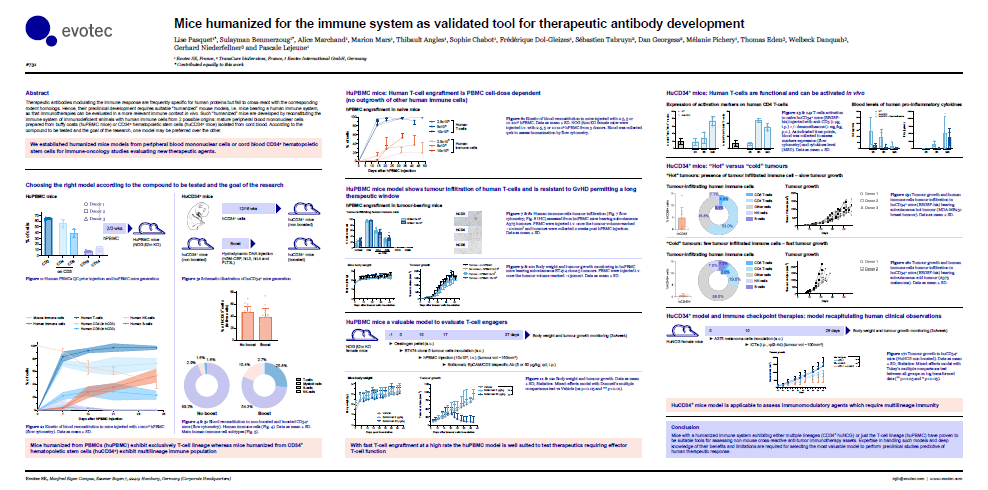
Novel preclinical immunocompetent mouse model for assessment of immunotherapies targeting cGAS-STING axis
Modulation of the cGAS-STING axis
Background: The cyclic GMP–AMP synthase–stimulator of interferon genes (cGAS–STING) signaling axis represents a pivotal immunostimulatory pathway and an attractive pharmacological target in oncology. Its activation within the tumor microenvironment promotes cross-priming of tumor-associated antigens and enhances infiltration of effector T lymphocytes. Owing to its potent anti-tumor activity, the cGAS–STING pathway offers significant promise for the development of cancer vaccines, immunotherapeutic strategies such as antibody-drug conjugates, and interventions against virus-driven malignancies. However, translating preclinical findings to clinical applications has been challenging due to species-specific differences between human and mouse cGAS and STING. A model expressing human STING only has been previously reported and showed to be a valuable tool to investigate the activity of STING agonist indifferent tumor types. Herein, we report an immunocompetent mouse model expressing both human cGAS and human STING (genO-hcGAS/hSTING).
Related products
Catalogue product

Scientific excellence
From model design to experimental results
Featured in 600+ scientific articles

Collaborative approach
Collaboration with 17 Top Pharmas,
170+ Biotechs and 380+ Academic Institutions

Robust validation data on catalog models
Generated with biopharma partners and in-house

Innovative technologies
and guaranteed freedom to operate

Easy access to models
Models with certified health status from professional breeders in US and Europe